The development of nanotechnology has been an exciting route to chemical innovation that has changed the way that we live. Fields as diverse as medicine, water treatment, and energy storage have all used nanoscale devices and nano-sized architectures to improve technologies related to human health, communications, and transportation. Nano is big business and projected to be even bigger in the next decade(s).
As the amount of nanotechnology in our daily lives increases, so does the need to understand the potential environmental impact and long-term fate of the nanoscale materials that we use. One pressing example is electronic waste, also called e-waste, from our mobile devices. The nanoscale materials in electronics can (in some circumstances) transform, releasing heavy metals and potentially toxic materials. That means that if e-waste is not properly disposed of at its end of life, a consequence could be unintended exposure to these heavy metals and other toxic materials. This could pose a significant risk to human health and the environment as the total amount of nanomaterials we use increases.

But how do these chemical transformations work? My colleagues and I recently published a research paper in which we used a computational method called density functional theory (DFT) to investigate how certain materials transform if they are exposed to water.1 Our specific example was a complex metal oxide material called NMC: Li(Ni1/3Mn1/3Co 1/3)O2. Because NMC (and related materials, like LiCoO2) are part of the batteries that power our mobile devices, they are projected to be a large component of e-waste in the future. We used computer modeling to figure out what happens to the surfaces of battery materials when they interact with water.
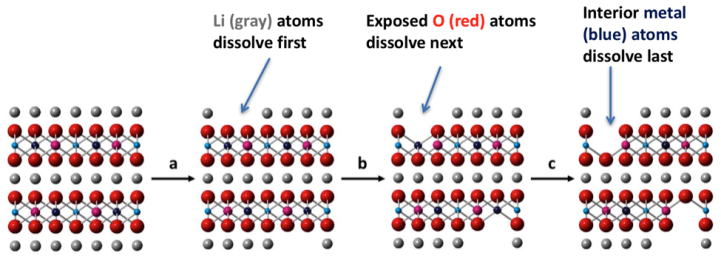
We found that in water, lithium (Li) will readily dissolve from the surface of NMC, and that under those conditions the surface of NMC would prefer to be covered with hydrogen atoms that it takes from water. We call this a hydrated surface, and used a combination of modeling and thermodynamics to show that once the surface is hydrated, it allows for the other metals to dissolve out of the material as well.
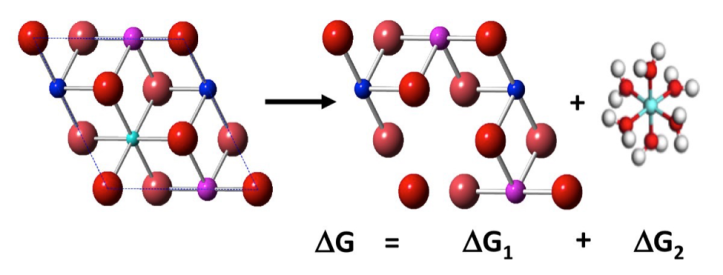
How did we use computer modeling to figure this out? We calculated a property called the free energy of dissolution, by combining well-known values based on experiments with our own DFT-calculated energies. An example illustration is shown in Figure 3 below. We were able to use basic equations (like the Nernst equation!) from high school chemistry class to include effects like changes in pH and concentration. We found that metals tended to dissolve out of NMC more readily at low pH (acidic pH of 3, like vinegar) than neutral pH (pH 6-7, like water), and that the dissolution was highly dependent on the number and location of different metals in the material. For example, manganese was more likely to dissolve from the surface of NMC when it was at 11% concentration than 17% concentration. This also means that the dissolution reactions stop after a while, because it gets to a point where too many metals have been removed, and it is not favorable for removing anything else from the NMC surface.
Different metals in NMC also dissolved at different rates. Overall, we found that lithium dissolves first, followed by nickel, cobalt, and manganese, which qualitatively matches with findings from previous benchtop experiments done by our Center for Sustainable Nanotechnology colleagues.
Our paper is an important step forward in connecting DFT calculations to experimentally measureable conditions for a couple of reasons. First, it allows us to use DFT to create rules for re-designing nanomaterials that are benign by design. It also yields insights into nanoscale transformations of complex metal oxides, an area that is important for understanding impacts on environmental health and safety. Future work will include how changing the composition of NMC will affect metal dissolution. For example, we will compare compositions that are more manganese-rich with others that are more nickel-rich. We hypothesize that metal dissolution is highly dependent on the identity and number of different neighboring metals, and that different amounts of metals will dissolve at different rates from different compositions. Complex metal oxides indeed!
REFERENCES
- Bennett, J. Jones, D., Huang, X., Hamers, R. & Mason, S. Dissolution of complex metal oxides from first-principles and thermodynamics: Cation removal from the (001) surface of Li(Ni1/3Mn1/3Co1/3)O2. Environmental Science & Technology, 2018, Available Online. Doi: 10.1021/acs.est.8b00054

Really?!
This article showed the depth of research and time that was pit to putting this up and trust me you did an awesome job at it.