Humans have been dreaming about space travel since the early ages. As many of you recall from the opening lines of Star Trek, it has been fascinating for humanity “to explore strange new worlds, to seek out new life and new civilizations, to boldly go where no one has gone before.”
However, space exploration involves many grand challenges including the extreme environments that the spacecraft has to tolerate: variations in gravity, solar radiation, and chemically different environments are all stumbling blocks that can greatly impact machine operations. Once the spacecraft is out of the atmosphere, it is also subject to incredible temperature changes not observed on Earth (thank you, atmosphere!). Even on Earth’s moon the temperature can range from about -250 °F (-157 °C) at night to +250 °F (+121 °C) with the sun hitting the surface.1

Due to the great temperature differences experienced during spaceflight, there has been significant research going on, predominantly at NASA,2 to develop insulating materials that can handle extreme temperatures. Fortunately, near the beginning of 20th century, technology finally caught up with imagination and a trip to space was no longer just a sci-fi dream.
As early as 1930, scientists developed a “mystifying blue smoke”3 that shined light into space explorations. This material, called aerogel (also known as “frozen smoke” or “solid air”), was first produced by dehydrating a gel while keeping the shape fixed. Imagine trying to remove the liquid from your Jell-O® dessert while keeping its shape — this is a hard task since it is about 85% water! When researchers were eventually successful in replacing the liquid phase of a gel with a gas they found that these aerogels had a structure similar to a kitchen sponge. But unlike a kitchen sponge, which has pores (pockets of air) on the order of millimeters, the aerogels have pores that are a million times smaller, on the order of nanometers.4

Today, there is a whole library of aerogels that have different compositions and exhibit different properties. Silica aerogels like the one above are the most common. It is composed of silicon dioxide particles (glass is made of the same compound) that are interconnected to form a porous network. The cloudy appearance of a silica aerogel comes from the nanoscale pores in its structure. To understand why, let’s remember how we see colors. Sunlight (and light emitted from a typical light bulb) is made up of all the colors of the rainbow and travels in waves. When light reaches a surface, it can bounce, be absorbed by the surface, or pass through it. When certain wavelengths bounce off and reach our eyes, we perceive the color of that surface based on those wavelengths. (For a more detailed reminder about this, see our post “What is Color and How Can It Help Kill Cancer Cells?”)
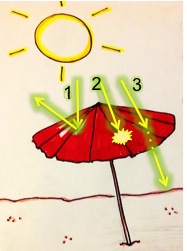
When light reaches an aerogel surface, it is scattered by the tiny pores, which are even smaller than the wavelength of light. Light with shorter wavelengths is scattered more since it is closer to the size of the pores. Blue light has shorter waves than red light, so blue is scattered more giving aerogel its blue color. This is called Rayleigh scattering and it is the same phenomenon that causes the sky to appear blue!5
Beyond their interesting interactions with light, aerogels exhibit other startling properties at the nanoscale. Aerogel is a porous material (like a sponge) where 97-99% of it is nothing but air. The air pockets in its structure serve as insulators since air does not conduct heat well. Materials that conduct heat well include metal, while air, wood, and rubber conduct it poorly. This is why you might use a metal coat-hanger to roast marshmallows, but put a wooden handle on the end to hold so you cook the marshmallow and not your fingers. Their low thermal conductivity is what makes aerogels good insulation materials.
Furthermore, since the solid framework only comprises 1-3% of the aerogel by volume while the rest is air, aerogels are very light materials. Today, a graphene aerogel holds the Guinness World Record for being the least dense solid on earth, which means it has the lowest mass in the large space it occupies!6 Yet, due to their solid framework, aerogels are strong materials if the force is uniformly applied.

Aerogels with these unique properties and seemingly endless “magical” capabilities have found major applications in space explorations:
Capture of cosmic dust
Aerogel was first used to capture interstellar dust particles from comet Wild 2 by the Stardust sample return mission.7 Collection and analysis of these frozen particles was important since they were expected to contain materials that formed our solar system. In order to collect samples, the Stardust Mission used a collector that consisted of two grids facing in opposite directions. The “tennis-racket” shaped metal framework contained 130 cells of silica aerogel of different volumes to create a density gradient between the grids.8 The high speed dust particles first hit the low density aerogel, and, as they penetrated through the material, the increase in density slowed the particles and eventually trapped them in the collector. This worked like a series of sieves or filters with progressively finer holes that the particles would pass through. The samples that were collected with minimal damage were then brought to Earth for further analysis.

Spacecraft Insulation
Since aerogels are such good heat insulators due to the air pockets within their porous network, they are used in spacecraft insulation. The aerogel is packed in composite boxes called “Warm Electronics Boxes” to protect batteries, electronics or computers from the extreme cold of deep space.8 After its successful use in the Mars Pathfinder mission, aerogel was employed in other Mars exploration rovers that can resist relatively large temperature fluctuations in the harsh environment of the Red Planet.

Containment of liquids at very low temperatures
With their excellent insulating properties, aerogels also have an important role in cryogenic fluid storage in space shuttles. Space shuttles burn liquid hydrogen and liquid oxygen as fuels. These are cryogenic fluids, meaning they have to be kept very cold: hydrogen must be stored at -423 °F (-253 °C) and oxygen must be kept at -297 °F (-183 °C) to remain liquid. Recently proposed aerogel formulations not only offer capabilities to be shaped into cylinders and trapezoids for transfer and storage of cryogenic liquids, but they also can be made to resist environmental corrosion.8
There are many other applications of aerogels in space exploration, including fire resistant space suits that provide insulation from the extreme environment and aerocapacitors that would use the high surface area of carbon aerogels to enhance energy storage devices.9
This technology is not only limited to space applications, but is also starting to be used for building insulation and outdoor apparel. You can now get a chic aerogel jacket for $300! OROS recently ran a Kickstarter campaign for aerogel gear to include jackets, gloves and snow pants.
Scientists from all around the world are working to improve the properties of aerogels. They are using different materials and different compositions to get stronger, more flexible, and lighter aerogels. Although aerogel technology is still limited by the high costs of production10 there is no doubt that these incredible materials with amazing properties will soon be more widely available in a product near you.
EDUCATIONAL RESOURCES
- NISEnet.org – Aerogel lesson (ages 7-adult)
- JPL Stardust – Technology for Studying Comets activity (grades 5-8)
- Benson, T. Moon, [NASA website]. 2014, retrieved from https://spaceflightsystems.grc.nasa.gov/education/rocket/moon.html
- Woods, T. Aerogels: Thinner, Lighter, Stronger. 2011, retrieved from http://www.nasa.gov/topics/technology/features/aerogels_prt.htm
- NASA/JPL. Aerogel Fact Sheet. JPL 400-1119 9/03.
- Aerogel.org. How is Aerogel Made? [n.d.] retrieved from http://www.aerogel.org/?p=4
- Erickson, K. Why is the Sky Blue? NASA SpacePlace [website]. 2016, retrieved from http://spaceplace.nasa.gov/blue-sky/en/
- Guiness World Records. Least Dense Solid. 2013, retrieved from http://www.guinnessworldrecords.com/world-records/least-dense-solid
- Whalen, A. Catching Comet Dust. Stardust: NASA’s Comet Sample Return Mission [website]. 2005, retrieved from http://stardust.jpl.nasa.gov/tech/aerogel.html
- Bheekhun, N., Talib, A., & Hassan, M. Aerogels in Aerospace: An Overview. Advances in Materials Science and Engineering, 2013, 1-18, doi: 10.1155/2013/406065
- Sasidharan, B., Kale, B., & Murugan, A. Inorganic materials for use in high energy density batteries in capacitors for electronic publications. In Bahadur, D., Vitta, S., & Prakash, O. (eds). Inorganic Materials, Recent Advances. New Delhi: Narosa Publishing House. pp 56-60.
- Technavio.com. Aerogel Market Definition and Growth Prospect. [website, n.d.] Retrieved from http://www.technavio.com/report/aerogel-market-2015-2019-global-trends-forecasts-vendor-analysis
Note: This post was edited on May 30, 2016 to correct a typo. -253 degrees Celsius equals -423, not -473 degrees Fahrenheit.
