As a fan of sci-fi, I recently watched two classic movies: the original Star Wars (which you’re probably familiar with) and Ghost in the Shell (a post-cyberpunk Japanese animation). Both movies had a very interesting commonality: human-machine hybrids. But what does this have to do with nanotechnology?

In the Star Wars movies, the only thing separating Darth Vader from death is his metal suit, which allows him to breathe and move. In Ghost in the Shell, almost all the characters have robotic implants. Some have electronics augmenting their brains while others have different levels of advanced prostheses. The main character, Motoko Kusanagi, even has a full-body prosthesis. That’s why the movie is called Ghost in the Shell: it is because an individual’s consciousness is wrapped in an artificial body and they can only communicate with the external world through this “shell.”

The machine-human hybrid is a common theme in science fiction, and there are many discussions challenging the boundary between humans and machines. Although the idea of a cyborg is still science fiction, science has brought forward a lot of fascinating developments at the inorganic-biological interface. In particular, my attention was captured by recent scientific progress in using nanoparticle-bacteria hybrids for solar energy capture.
We are so familiar with the sun, we often don’t even think about it. But not only does the sun provide us with heat, but the food we eat everyday ultimately comes from the sun too. Plants can capture sunlight to produce chemicals that can store energy in a process known as photosynthesis. The nutrients from these plants can then be digested by livestock and humans and used as energy. Thus, in today’s world where food security is a big deal and fossil fuels are slowly being depleted, photosynthesis means a lot to all of us.
However, natural photosynthesis has its downsides. Photosynthesis is mainly composed of two processes:
- a light harvesting process to convert energy from sunlight to electrons, and
- a series of electrochemical reactions that uses these electrons to form new chemicals

Just like how human brains cannot exceed the calculation speed of a computer, the light harvesting efficiency of a plant cannot compete with that of an inorganic semiconductor. On the other hand, nothing artificial is currently better than what occurs in nature to carry out the second process of photosynthesis (forming new chemicals) because of the catalytic power of biological enzymes. Enzymes are (mostly) proteins that accelerate the chemical reactions in cells. Enzymes are so good at what they do that currently, nothing human-made can compare to this beautiful natural system.
What if we could combine the light harvesting capability of inorganic semiconductors and the catalytic power of biological cells? Can we have a science-fiction-style hybrid where robotic devices enhance their natural abilities?
Scientists have tried this “what-if” in what they call “artificial photosynthesis” (luckily there’s no ethical problems in making bacterial “cyborgs”!) In this cutting-edge research, nanomaterials have played a critical role. Nanoscale semiconductors known as quantum dots can absorb light of certain wavelengths based on their sizes. A previous Sustainable Nano blog post described these materials in detail. The excellent light-harvesting capability of these nanoscale semiconductors make them ideal for the development of artificial photosynthesis, as these quantum dots absorb light even better than the light-absorbing green pigments in plants.

Recently, scientists in Berkeley, California were able to make a nano-bio hybrid system from a non-photosynthetic bacterium and nanoscale cadmium sulfide (CdS) quantum dots.1 This hybrid can use light and carbon dioxide from air to produce acetic acid. Acetic acid is the main component of vinegar besides water. So essentially, this hybrid is using carbon dioxide from air to make vinegar!

Interestingly, these CdS nanomaterials were not completely foreign to bacterial cells. You might think this experiment would require putting manufactured nanomaterials and bacterial cells together to form a hybrid. The same researchers did do that in some earlier work,2 but in this case these CdS nanomaterials were actually made by the bacteria themselves when proper chemicals were added. As you can see in the beautiful electron micrograph shown below, the exterior of bacterial cells were covered with a “shell” of CdS nanomaterials. That’s why I call it “Bacteria-in-The-Shell”—it reminds me of how a human consciousness was put into a robotic shell in Ghost in the Shell.
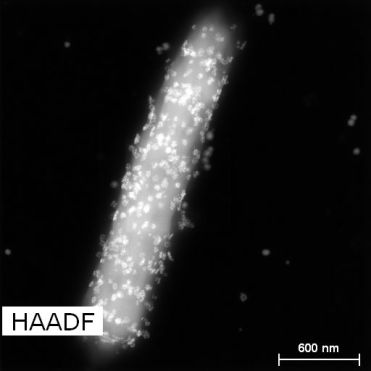
The CdS shell in this case acts as a light-harvesting factory for the first step of photosynthesis. When lights hits these CdS nanomaterials, electrons absorb the light, get excited, and jump to a higher energy level. These excited electrons then initiate a series of biochemical reactions with the help of enzymes in cells to finally produce acetic acid. In this nano-bio hybrid, the abiotic and biotic parts work closely with each other to produce useful chemicals and keep the bacterial cells alive. Without light or the nanoparticle shell, these bacterial cells died quickly. I would say that this is the most fascinating part of the hybrid system!
Such integration of abiotic and biotic components is just like what people have dreamed about in science fiction. In Ghost in the Shell, everyone has cybernetic parts in the year 2030. Maybe we are not going to all become cyborgs by that time (though I would love to), but the technology is already on its way for such nano-bio hybrid systems to help us harvest more energy from the sun and nourish our world.
EDUCATIONAL RESOURCES
- Science & Plants for schools book: A Leaf in Time – A Popular Introduction to Photosynthesis (middle school)
- NOVA interactive Illuminating Photosynthesis
- Sciencegeek.net: “Toothpickase” enzyme activity
REFERENCES
- Sakimoto, K., Wong, A., & Yang, P. Self-photosensitization of nonphotosynthetic bacteria for solar-to-chemical production. Science, 2016, 351 (6268), 74-77. DOI: 10.1126/science.aad3317
- Liu, C., Gallagher, J., Sakimoto, K., Nichols, E., Chang, C., Chang, M., & Yang, P. Nanowire–Bacteria Hybrids for Unassisted Solar Carbon Dioxide Fixation to Value-Added Chemicals. Nano Letters, 2015, 15 (5), 3634–3639. DOI: 10.1021/acs.nanolett.5b01254
