This post is part of our ongoing series of public-friendly summaries describing research articles that have been published by members of the Center for Sustainable Nanotechnology. Vivian Feng, a chemistry professor at Augsburg College, is the first author of this paper, which was a collaboration with undergraduate and graduate students as well as other CSN faculty members. Vivian says, “Understanding the impact of nanomaterial toxicity on the environment is a highly interdisciplinary task. I really valued the team effort while working on this manuscript, especially for my undergraduate students to experience it. The boundaries between disciplines of chemistry, biology and physics were blurred when we all put our heads together to solve the same problem – that’s the beauty of a collaboration.”
The article was first published online in June 2015 in the journal Chemical Science.1
“Impacts of Gold Nanoparticle Charge and Ligand Type on Surface Binding and Toxicity to Gram-Negative and Gram-Positive Bacteria”
If you’ve ever gotten sick from a bacterial infection, you are probably not a stranger to a special class of chemicals, commonly known as “antibiotics.” These powerful drugs have helped many people fight bacterial infections from simple strep throat to life-threatening diseases. In recent years, researchers have discovered that various types of nanomaterials demonstrate great antibacterial properties. Hence an increasing number of commercial products incorporating nanomaterials are marketed with an “antibacterial” label. (Want to know more? Check out our post, “Silver for your smelly socks“!)
Killing odor-inducing bacteria in stinky socks may be viewed as a heroic action for nanoparticles, but are all bacteria bad? What about those hardworking bacteria that help break down organic matter in soil to provide nutrients to plants? Since we don’t want to kill all bacteria indiscriminately, we need to be able to reduce how much nanomaterials affect beneficial bacteria when released into the environment. In order to do that, we need to understand exactly how nanoparticles interact with bacteria in the first place.
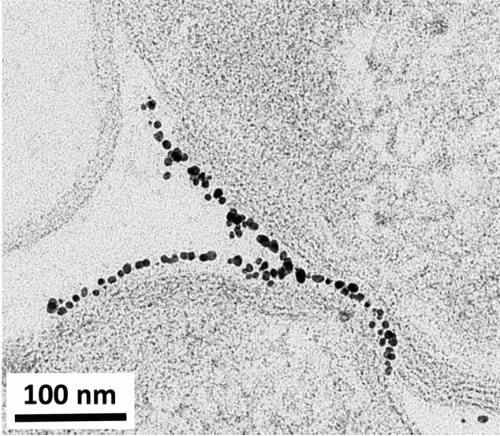
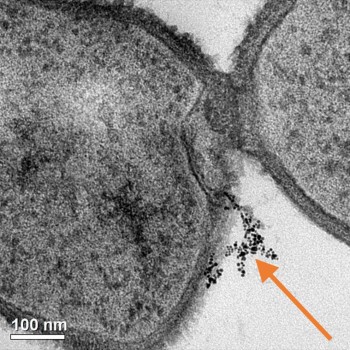
Although both bacteria and nanoparticles are tiny entities, we need to take a dive even deeper and examine the actual “points of action” on their surfaces. In the study described in our new article, we investigated the significance of both the surface of the bacteria and the surface of the nanoparticles. We systematically evaluated two types of bacteria with different cell surface chemistry (Shewenella oneidensis and Bacillus subtilis) and three types of gold nanoparticles with different surface chemistry. (See this post for more about Shewanella.)
One big difference between Shewanella and Bacillus bacteria is that Shewanella has cell walls that feature a sandwich structure with two lipid bilayers with a polymer network between them, and Bacillus has a simpler cell wall structure with just one lipid bilayer underneath a thick layer of polymer network. When scientists study cell membranes, they use something called a “gram stain,” a dye that turns sandwich-style membranes like Shewanella’s pink, and simple membranes like Bacillus’s purple. Based on these stains, we say that Shewanella is a “gram negative” bacteria, and Bacillus is a “gram positive” bacteria. Because of this contrast, and because both bacteria are environmentally beneficial, we chose them as the representative gram negative and gram positive bacteria to test with nanoparticles in our study.

Something these two types of bacteria have in common is that they both have a negative charge on their surfaces. Based on electrostatic interactions (i.e. opposites attract), we hypothesized that gold nanoparticles with a negative charge on their surface would not be attracted to bacterial cell surfaces, meaning that they would have minimal impact on whether the bacteria died. In contrast, we predicted that positively charged nanoparticles would bind strongly to cell surfaces, which could then cause cell death. Based on this hypothesis, we tested one type of gold nanoparticle with a negative surface, one with a positive surface, and one with a polymer wrapping on the surface that gave it an even higher positive charge per square nanometer.
So, did our experimental results support our hypothesis? Using several techniques including transmission electron microscopy (which we wrote more about here), we found that positive and negative charges indeed governed the interactions between these nanoparticles and the cells. Even more interestingly, we not only showed that higher surface interactions led to more cell death, but we also demonstrated that the different cell surface features of the two types of bacteria led to different kinds of binding behaviors with the nanoparticles.
Now that we understand a little bit more about how different bacterial cell surfaces interact with different kinds of nanoparticles, we are one step closer to figuring out how to redesign nanomaterials so that they are exactly as antibacterial as we want them to be, and no more.
EDUCATOR RESOURCES
- BrainPOP Virtual lab: Gram Staining
- Nuffield Foundation/Society of Biology class practical: Investigating anti-microbial action
REFERENCE (full article is available open access)
1. Feng, V.Z.*, Gunsolus, I.L., Qiu, T.A., Hurley, K.R., Nyberg, L.H., Frew, H. Johnson, K.P., Vartanian, A.M., Jacob, L.M., Lohse, S.E., Torelli, M.D., Hamers, R.J., Murphy, C.J. & Haynes, C.L.* Impacts of gold nanoparticle charge and ligand type on surface binding and toxicity to Gram-negative and Gram-positive bacteria, Chemical Science, 2015, Advance article, doi: 10.1039/C5SC00792E
Note: This post was edited August 18, 2015 to replace “common cold” with “strep throat” as an example of a common bacterial infection. Common colds are caused by viruses, not bacteria!

[…] Vivian Feng mentioned in her recent post, there are two ways that bacteria are likely to encounter nanomaterials: when the bacteria are […]