The idea that an image is worth a thousand words applies just as much in science as it does in other contexts. Using images, scientists can convey complex information more quickly and often more clearly than is possible using words—which do you prefer: words (left) or a picture (right)?
Today I want to talk about how we in the Center for Sustainable Nanotechnology zoom in on the nanoworld; specifically, how we see living things, like cells, interacting with nanomaterials. We care about this because we think that nanomaterials, given their incredibly small size, may interact with living things in strange ways. But if we want to see this interaction, we’ll first need to come up with some way to see things on the nanoscale. It turns out that this is no easy task, which may not surprise you. But why exactly is it so hard to see small things like nanomaterials and cells? And how can we overcome these difficulties?
One idea is to use a microscope. Microscopes are used all the time to observe things that can’t be seen with the unaided eye, and they accomplish this by using a series of lenses to produce a magnified image of a sample. Unfortunately, it turns out that normal microscopes, like the ones you may have used in biology class, aren’t quite good enough to see nanomaterials. Part of the reason is that when light passes through a lens (like the lenses we use in a microscope) it spreads out a little bit. This is known as diffraction.
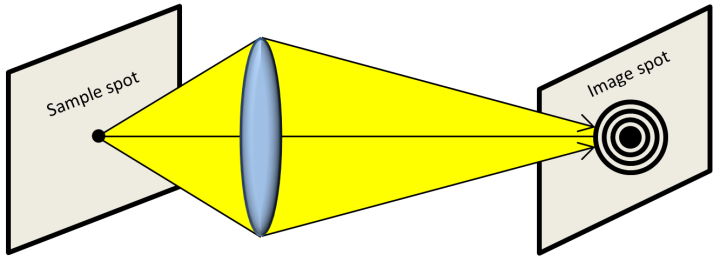
Let’s say that we want to take an image of the spot on the left in the above image using a microscope. Microscopes are fairly complicated, so let’s simplify our picture by imagining that our microscope only has one lens. We collect light bouncing off our spot using this lens, and adjust things until we make an image that’s in focus on the right. Unfortunately, the image that we create looks a bit more spread out than the original spot. Why is this? It turns out that when light passes through a lens it spreads out a little bit; this is known as diffraction, and the spread out pattern of our image is called an Airy disk. Just imagine that there are two nearby spots that we want to see (for example, a nanoparticle stuck to the wall of a cell). When we take an image of these spots, they look to us like two Airy disks—they appear a bit bigger than they should. This means that we won’t be able to tell the spots apart if they are close enough together that their Airy disks overlap too much. This is the problem that we run in to when we try to see nanomaterials in a normal microscope. Diffraction is limiting us from seeing really small things really close together—scientists refer to this as the diffraction limit.
So how can we get around this? Fortunately, there are some tricks that we can use to zoom in on cells and see how they interact with nanomaterials.
The general term for the clever tricks that we can play with light to break the diffraction limit and see really small things is super-resolution fluorescence microscopy. There are a few different types of this microscopy, but today I want to talk about just one, called stochastic optical reconstruction microscopy (abbreviated STORM). Recently, I’ve been working on using this recently invented technique to take images of nanomaterials interacting with bacterial cells. This work is still in progress, but to see what STORM can do, check out this image of tiny cellular skeleton components known as microtubules. These play a critical role in maintaining cell shape, moving substances around within a cell, in addition to cell division processes.

If we were using a regular microscope, we would have no hope of seeing the detail that we can see in this STORM image. So how does STORM do this? What’s the trick?
First, our sample needs to be fluorescent. This means that when we shine light on our sample, it needs to emit light back (for more on this, see Tom’s previous post on fluorescence). We also need this emitted light to turn on and off over and over again while we take an image. This is the hard part of STORM—finding a way to make our sample turn its light on and off. I’ve been working on this a lot recently, and will talk in a future post about what I did to make bacteria emit light in this special way.
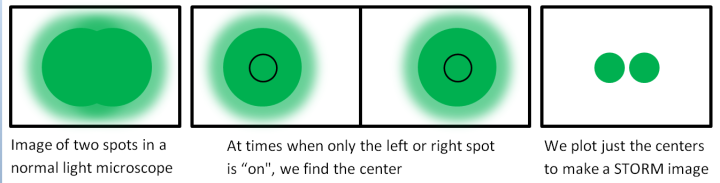
Once we have a sample that can turn its light on and off repeatedly, we are ready to do STORM. Let’s say that want to take images of two spots in our sample that are really close together. Maybe the spot on the left is a cell and the spot on the right is a nanoparticle. We shine light on them and record lots of images as the emitted light from each spot turns on and off. In a normal light microscope, the images of the spots overlap due to diffraction and we can’t tell them apart (image below, left). But in our STORM microscope, there are times when only one of these spots was “on” while the other is “off”. If we collect lots of images, we’ll find cases where only one spot is on and not the other (image below, middle). Now that the neighboring spot is out of the way, we can find the center of each spot and make a new image with just the centers (image below, right). By doing this, we can distinguish the two spots—we can see the cell and the nanoparticle!
Right now, our Center is using STORM and other kinds of tricks to see the interactions of nanomaterials with living things. We think that by seeing this interaction, we can better understand how nanomaterials behave. Stay tuned to see some of our exciting results!


[…] chest of fluorescence microscopes capable of performing super-resolution techniques such as STORM, SIM (see a SIM tutorial here), and single molecule tracking, which are some of the same […]
[…] Here at Sustainable Nano, we’ve published several posts talking about the instrumentation used by researchers in the Center for Sustainable Nanotechnology to visualize nanoparticles and cell membranes, such as atomic force microscopy and super-resolution fluorescence microscopy. […]
[…] the world of molecules with ease, let me mention some of the limitations of these techniques. We discussed in a previous post how the diffraction of visible light limits us from resolving (or distinguishing) objects less […]
[…] determine the center of the location of each blinking spot. Ian described this method in more detail in another blog post. For comparison, a traditional light microscope allows a viewer to distinguish objects as small […]
[…] a nano-crisis One of the Most Sensitive Balances on the Planet – Quartz Crystal Microbalance Zooming in on the Nano World: Limitations and Breakthroughs in Light Microscopy Surfing Particles: taking a ride using living cell machinery The Canary in the Coal Mine and a […]
[…] In water the nanoparticles are clear, but we can intentionally make them fluorescent. That way when we shine light of one color on the diamond nanoparticles they will give off light of a different color. Even if the nanoparticles are too small to see by eye, we can still track them using the light they emit. […]
[…] in an object are too close, their image disks overlap, and we cannot see the two unique features (see Ian’s post for more). When the resolution is only limited by diffraction, the distance needed between objects in order […]
I am doing a project on a nano bathing suit. That is one of the things that you can find nano technology in. I have been trying to contact a scientist that knows of the nano quick drying bathing suit. Do any people on this site know of a scientist, or even maybe is a scientist that knows of or has researched this? Thank you for all of your responses on the other blog.
Hi Nano_loverrr510!! Awesome question! Luckily a scientist in our lab, Sam Lohse, has responded to a similar question on another post. Check out his response here:
https://sustainable-nano.com/2013/10/28/hewitt-school-adventures-in-the-nano-zone/comment-page-1/#comment-4481
The video he links to there is super awesome.
If things are clothes are drying, that means water is repelled by the surface of the fabric, so the fabric never really gets “wet” like other fabric. Scientists call this “hydrophobic”. There is such a thing as “superhydrophobic” (more in the video in Sam’s comment).
Check out this link: http://www.nanowerk.com/news/newsid=9364.php
Unfortunately it’s difficult to know exactly what nanomaterials the manufacturers are using because they are not required to release that information. In many cases this is because the manufacturers invested a lot of money into developing their special nanomaterial and don’t want anyone else to steal their idea. But, we can definitely say in this case that it has something to do with a superhydrophic coating that the manufacturers claim takes advantage of nanotechnology. Hope that helps! Thanks for your question!