If you’re like me, you often find yourself thinking about random things. For example, just the other day I was thinking to myself, “what’s the diameter of a gold nanoparticle* in light years**?” Unit conversions have been a thorn in my side for as long as I can remember, so I was pleased to discover one of the coolest Google features ever: unit conversions! (Just when I thought Google had maximized my laziness…)

So, with Google’s help, we can figure out the diameter of a gold nanoparticle in light years. We’ll assume our gold nanoparticle is 10 nanometers in diameter (similar in size to many of the nanoparticles we use in the Center for Sustainable Nanotechnology – check out some previous Sustainable Nano posts to learn more). All you have to do is type the following into your Google search bar: “10 nanometers in light years” – go ahead, I’ll wait.
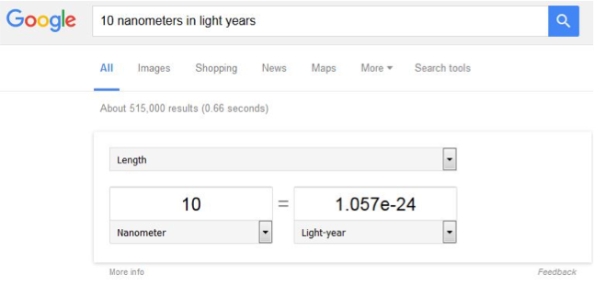
The result you see on your screen is 1.057e-24, which is a different way of writing 1.057 x 10-24, which is scientific notation for 0.000000000000000000000001057. This really small number is the diameter of our gold nanoparticle in light years!
This exercise got me thinking about how the numbers that scientists use are often really hard to fathom. For example, the Milky Way Galaxy is approximately 100,000 light years across, or 600,000,000,000,000,000 miles (that number is 600 quadrillion miles, or 9.7 x 1029 nanometers). Meanwhile, teeny tiny atoms are approximately one angstrom in diameter (0.1 nanometers, which is one ten billionth of a meter or 1.057 x 10-26 light years).
How can we conceptualize these vastly different scales? One way is to make random (and sometimes goofy) comparisons using objects that are a little easier to comprehend. For example, to put the size of an atom in perspective, our 10 nanometer diameter gold nanoparticle contains about a quarter million gold atoms (see how to calculate that number in a recent Sustainable Nano blog post), and hundreds of those nanoparticles would barely cover the tip of a mite’s piercing-sucking mouthparts (yup, they’re really called that)! The mite isn’t very big, either; it could hide in the king’s crown on a gold coin that’s a little smaller in diameter than a penny.
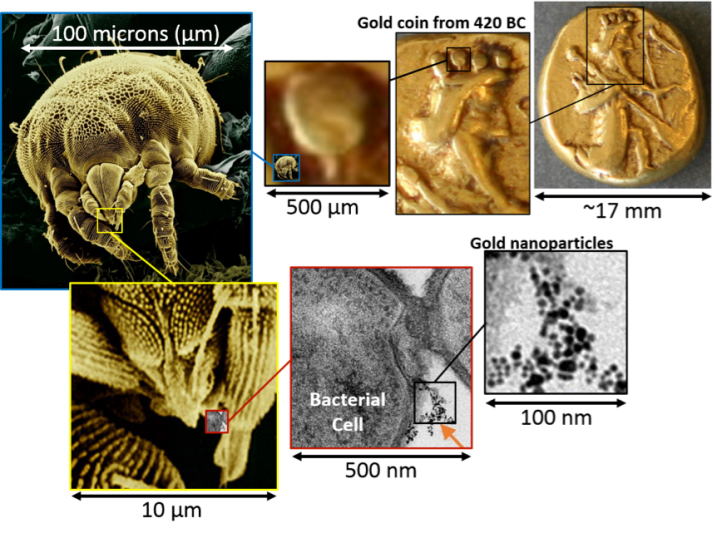
But, as much fun as it is comparing nanoparticles to gold coins and the business end of creepy-crawlies (see this amazing ladybug zoom-in post if you want more), what really makes science fun is actually doing the experiments, which means measuring things! Here at the Center for Sustainable Nanotechnology we love to measure tiny things like nanoparticles – it’s what we do! You can find out more about how we measure really small objects in these blog posts about cool instruments like microscopes, lasers, and more.
And we’re not the only ones using cool instruments to measure things. Have you heard of the Large Hadron Collider (or “LHC”)? The LHC is the most advanced scientific instrument ever built, and it’s used to smash protons into each other at almost the speed of light so that scientists can study some of the most basic questions in physics, such as the behavior (or even the very existence) of elementary particles (the building blocks for protons, neutrons, and electrons found in atoms)!
While the Large Hadron Collider is a very big instrument (the circular tunnels have a circumference of about 27 kilometers, or 17 miles), the colliding protons are really, really, really tiny – with a radius less than a femtometer (a femtometer is 1 million times smaller than a nanometer!). Put another way, if our gold nanoparticle was Earth, a proton would be about the size of a human hamster ball!

Speaking of planets, scientists like to measure big stuff too, which can get a little tricky when the thing you want to measure is also very far away. For instance, back in 2006, NASA launched the New Horizons spacecraft to conduct a reconnaissance of Pluto – a trip that took almost 10 years and covered about 3 billion miles! The journey took so long that Pluto was a planet when New Horizons was launched, but a dwarf planet when the spacecraft finally arrived – plus, three of Pluto’s moons were discovered along the way (that’s a long trip)!

While a precise understanding of gravity isn’t critical when you want to roll a human hamster ball down a hill, it becomes very important if you want to calculate the paths of both a spacecraft and a planet so they end up in the same place at the same time. Fortunately, Galileo began rolling balls down hills to change the way we think about gravity more than 400 years ago (though his notes make no mention of human hamster balls), and NASA has been taking advantage of that knowledge since a time when landing spacecraft on the moon required using slide rules!
In fact, NASA has gotten so good at measuring gravity that scientists were able to get the New Horizons spacecraft to Pluto a little faster by using Jupiter’s gravity as a slingshot (providing another nice photo op). To make the swing around Jupiter, the piano-sized New Horizons had to hit a window 500 miles across – “the equivalent of shooting a skeet target in Baltimore, Maryland from a firing range in Washington,” according to NASA scientist Alan Stern.
Needless to say, scientists have to take their measurements very seriously if they want to do amazingly cool things like smash protons together or send a spacecraft on three billion mile journeys to take pictures of former planets and their moons. Scientists take their measurements so seriously, in fact, that there is an actual agency tasked with making sure we’re all using the same units. It’s called the International Bureau for Weights and Measures. To learn more about units and the mind boggling ways they are defined, stay tuned for part 2 of this post, coming next week!
FOOTNOTES
*As you may know from previous Sustainable Nano blogs, we study gold nanoparticles in the Center for Sustainable Nanotechnology, and the “nano” in nanoparticle (or nanotechnology) refers to the size scale – a nanometer equals 1 billionth of a meter (in other words, there are 1 billion nanometers in a meter).
**A light year is a measure of length, not time – it’s the distance light travels in one year – and it’s a big number. Light travels at 186,000 miles per second (that’s 299,792,458 meters, or 299,792,458,000,000,000 nanometers in a second!). So, in one year, light travels 186,000 (miles per second) x 60 (seconds in a minute) x 60 (minutes in an hour) x 24 (hours in a day) x 365 (days in a year) = 5.865696 x 1012 miles, or 5,865,696,000,000 miles (that’s almost 5.9 trillion miles).

[…] my last post, I told you about how scientists measure amazingly cool things that can be vastly different in […]
This article is hilarious as well as informative!